Orphan drug developers face a temperamental FDA and must battle for investor attention with blockbusters in obesity and other common diseases. However, according to Evaluate’s 2026 Orphan Drug Report, global sales of orphan drugs are projected to grow and exceed $400 billion by 2032.
The annual revenue from rare disease treatments is projected to reach $409 billion in 2032, representing one-fifth of the nearly $1.9 trillion in prescription drug sales. This is a 15% increase from 2022, according to a new report from Evaluate.
The outlook is optimistic, but the path to $400 billion will not be straightforward. The market remains vulnerable to increased FDA scrutiny and potential pricing pressures. By 2032, ten existing drugs will lead the list of best-selling orphan drugs, with eight of them forecast to generate over $6 billion annually each.
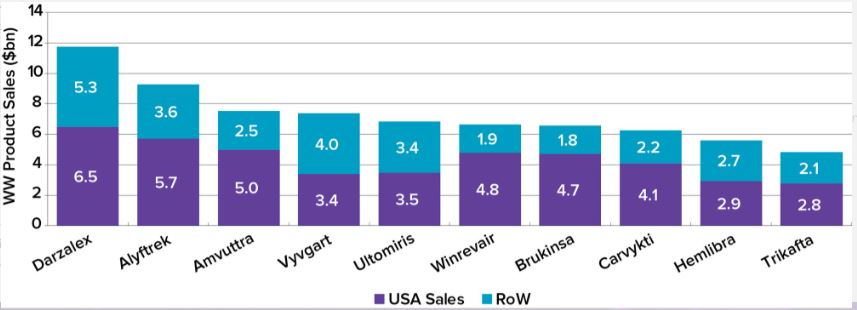
Leading this race is Johnson & Johnson, whose Darzalex (daratumumab) for multiple myeloma is set to become the company’s top-selling orphan drug with sales of $11.8 billion. Although this is lower than the $14.4 billion recorded in 2025, its subcutaneous formulation extends its market exclusivity, effectively doubling the franchise’s commercial lifespan. J&J’s position is further strengthened by Carvykti, developed in partnership with Legend Biotech for multiple myeloma, which ranks eighth with projected sales of $6.3 billion.
Thanks to these drugs, J&J is poised to claim the title of the largest company by orphan drug sales, forecasting revenue from this category at nearly $31 billion in 2032, the Evaluate report states.
Another company with two drugs in the top ten is Vertex. Alyftrek, a once-daily triple combination therapy for cystic fibrosis, is on track to become the world’s second best-selling orphan drug, with projected sales of $9.3 billion by 2032. Their older drug, Trikafta, will rank last with sales of $4.9 billion.
Alnylam’s RNA interference therapy for transthyretin amyloidosis (ATTR), Amvuttra (vutrisiran), is forecast to take third place, reaching sales of $7.5 billion by 2032. However, some questions surround the growth potential of this drug and the entire ATTR market due to the anticipated loss of market exclusivity for Pfizer’s first-in-class drug, tafamidis.
The most notable change in the rankings is the rise of Argenx. According to forecasts, it will displace Pfizer from the leaders in orphan drug sales in 2032, driven by the successful launch of its autoimmune drug Vyvgart (efgartigimod alfa). The projected total sales of $11.2 billion place the Dutch biotech company in eighth place, ahead of Merck & Co. ($9.9 billion) and Bristol Myers Squibb ($9.7 billion). Argenx is also the only company in the Evaluate list whose entire 2032 revenue is derived from orphan drug sales.
Other top orphan drugs include AstraZeneca’s complement inhibitor Ultomiris, Merck’s pulmonary arterial hypertension drug Winrevair, BeOne Medicines’ blood cancer drug Brukinsa, and Roche’s hemophilia A therapy Hemlibra.
Top 10 Orphan Drugs by Projected Sales in 2032
Merck & Co. can boast the fastest growth in orphan drug sales at 23% annually from 2025 to 2032, followed closely by Argenx with 22%.
In contrast to the overall growth of orphan drugs’ share in the pharmaceutical market, their presence in the R&D pipeline is beginning to decline. Evaluate analysts expect the share of pipeline drugs for rare diseases in total pharmaceutical sales to drop from a projected peak of 30% in 2027 to 22% in 2032.
“This likely reflects growing interest in investment in the development of other drugs, primarily those based on GLP-1,” noted the Evaluate analysts. As big pharma faces a total of $300 billion in patent expiries by the end of the decade, the need to develop drugs for a broad range of indications remains high, they added.
J&J’s Darzalex and Argenx’s Vyvgart owe their strong sales forecasts in large part to a recent amendment to the Inflation Reduction Act (IRA). Under the original IRA, only drugs with a single orphan indication were exempt from price negotiations with the US government. Donald Trump’s One Big Beautiful Bill Act expanded this to include drugs with multiple indications.
Global Orphan Drug Sales (2025/2032): Top 10 Companies and Total Market
However, the regulatory environment has recently become unpredictable for rare disease drug developers, a situation Evaluate attributes to “mixed signals” from the FDA.
The agency has introduced mechanisms to accelerate the development of personalized treatments targeting the genetic abnormalities underlying rare diseases, but simultaneously, the FDA has rejected several rare disease drugs, sparking industry backlash. The agency refused to approve Regenxbio’s gene therapy for Hunter syndrome and rejected Biohaven’s troriluzole for spinocerebellar ataxia.
Biohaven’s director, Vlad Coric, condemned a “systemic problem” at the FDA and warned of a “difficult period” for patients with rare diseases.
Furthermore, the rare disease market remains largely untapped, as current orphan drugs address only about 5% of rare disease cases, the Evaluate report notes.